Selank is a synthetic heptapeptide engineered by appending a Pro-Gly-Pro stabilizing tripeptide to the C-terminus of tuftsin (Thr-Lys-Pro-Arg), an endogenous tetrapeptide. This molecular architecture was developed to improve the metabolic durability of the tuftsin backbone while maintaining its bioactivity, making Selank a useful tool for laboratory investigations into neuropeptide signaling and neuroimmune communication.
The compound has attracted considerable attention in neuropharmacological research. In vitro work conducted on neuronal cell cultures indicates that Selank influences the expression of brain-derived neurotrophic factor (BDNF) and nerve growth factor (NGF), along with their associated receptor tyrosine kinases TrkB and TrkA—findings that have made it relevant to research into neurotrophic signaling cascades. Additional published studies have explored how Selank affects the expression of enkephalin-degrading enzyme genes as well as components of the GABAergic and serotonergic systems, positioning it at the intersection of neuroimmunology and neuropeptide biology. Its comparatively stable profile relative to native tuftsin supports use in longer-duration in vitro experimental protocols.
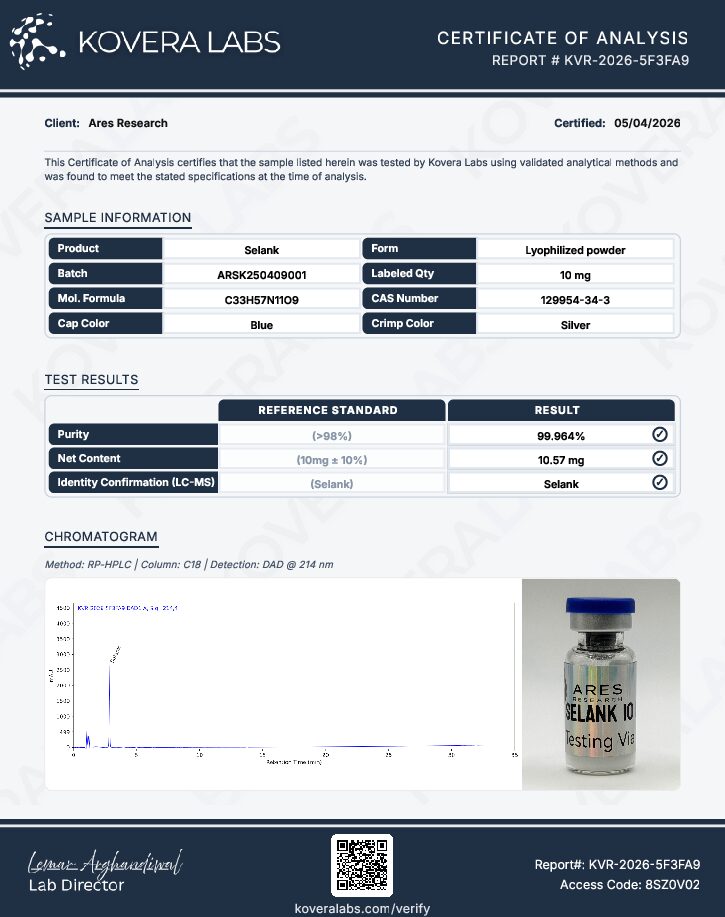
Ares Research supplies Selank as a research-grade lyophilized peptide accompanied by analytical documentation. This product is intended exclusively for laboratory research purposes and is not for human or veterinary use and has no intended clinical application.
FDA Disclaimer
The information presented on this website has not been evaluated by the U.S. Food and Drug Administration. No content on this site, nor any product offered, is intended to diagnose, treat, cure, or prevent any disease or medical condition.
All products are supplied solely as laboratory research reagents and are not for human consumption. Ares Research does not supply products to patients and makes no representations regarding therapeutic or medical use.
Ares Research operates as a chemical supplier only. It is not a compounding pharmacy and does not function as a compounding facility under Section 503A of the Federal Food, Drug, and Cosmetic Act.