Semax is a synthetic heptapeptide analog of the adrenocorticotropic hormone (ACTH) fragment 4-10, bearing the sequence Met-Glu-His-Phe-Pro-Gly-Pro. The C-terminal Pro-Gly-Pro extension functions as a metabolic stabilizer, conferring enhanced resistance to aminopeptidase and carboxypeptidase degradation and extending the effective experimental window in cell culture and tissue preparation studies. These properties have made Semax a well-established reagent in neuroscience research for investigating neurotrophic and neuroprotective signaling mechanisms across multiple model systems.
Published studies have documented the effects of Semax on BDNF and NGF expression in hippocampal and cortical neuronal cultures. Additional investigations have examined its influence on dopaminergic and serotonergic neurotransmission, modulation of immediate early gene expression including c-Fos and c-Jun, and effects on long-term potentiation (LTP) in electrophysiological assay systems. The compound has also been studied in models of oxidative stress, excitotoxicity, and neuroinflammation, positioning it as a versatile tool for researchers exploring neuroprotective signaling cascades and their molecular determinants in neuronal cell populations.
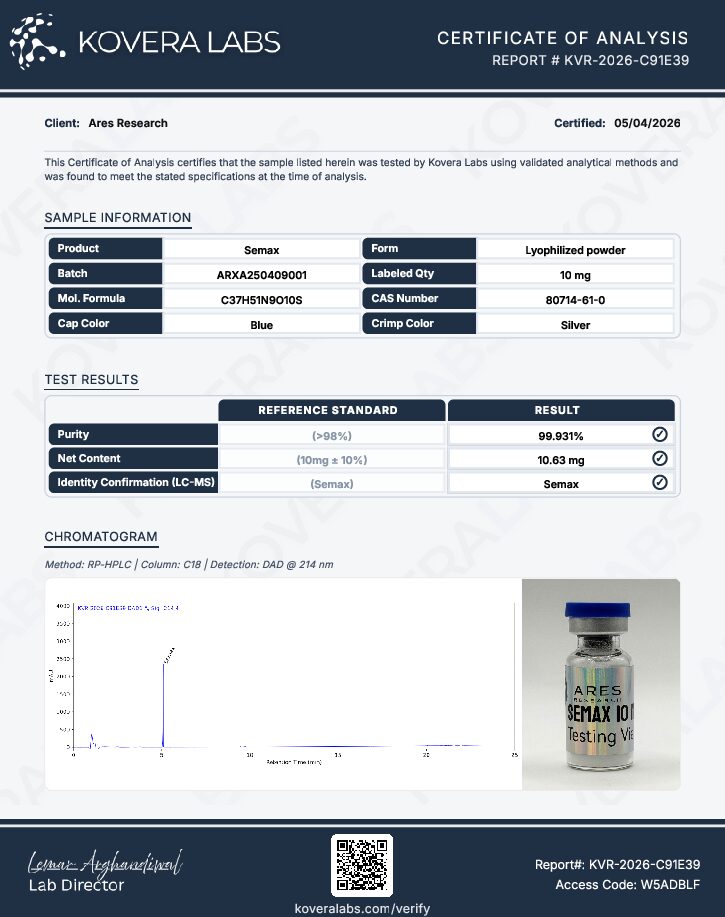
Ares Research supplies Semax as a research-grade lyophilized powder with documented purity and identity. This product is provided exclusively for in vitro laboratory research and is not approved for human or veterinary use.
FDA Disclaimer
The information presented on this website has not been evaluated by the U.S. Food and Drug Administration. No content on this site, nor any product listed, is intended to diagnose, treat, cure, or prevent any disease or medical condition.
All products are supplied solely as laboratory research reagents and are not for human consumption. Ares Research does not supply products to patients and makes no representations regarding therapeutic or medical use.
Ares Research operates as a chemical supplier only. It is not a compounding pharmacy and does not function as a compounding facility under Section 503A of the Federal Food, Drug, and Cosmetic Act.